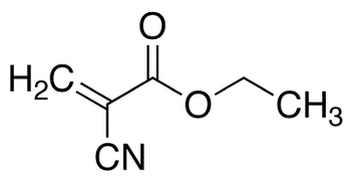
The Chemical Structure
Super glue is a very specific type of glue made of the ingredient cyanoacrylate. Cyanoacrylates are a family of strong fast acting adhesives. This family includes methyl 2-cyanoacrylate (super glue), ethyl 2-cyanoacrylate (krazy glue), n-butyl cyanoacrylate and 2-octyl cyanoacrylate (used in medical applications), etc...
|
Super glue exists as a liquid form consisting of monomers of cyanoacrylate molecules. These basic monomer structures are important for the bonding process. Also, due to the fact that cyanoacrylate is a polar molecule, it is hydrophilic, thus allowing it to bond and interact with other polar molecules. This trait attributes to the glues ability to strongly bond with molecules like the hydrogen in your skin.
One of super glues most feared enemies is temperature, which has the ability to make our hero, super glue, rigid and unyielding . How does temperature overpower our hero? Well, at a certain temperature, known as the glass transition temperature, glues becomes malleable. This temperature is dependent on the monomer side groups: the more flexible the group, the lower the temperature needed to defeat our hero. Each of our heroes must battle a different version of the villainous temperature and this coupled with the difference in their side groups is what allows for the different isomers of cyanoacrylate. As you can see in Figures 1 and 2, ethyl cyanoacrylate has one more carbon on the far right side of its structure than methyl cyanoacrylate. |